Uganda to launch HIV-prevention drug Lenacapavir on March 25
The categories of people to benefit include adolescent girls and young women aged 15-24, pregnant and breastfeeding mothers, female sex workers, and their male clients. The other targeted populations include fishermen, long-distance truck drivers, and people in multiple sexual partnerships.
The national pre-exposure prophylaxis (PrEP) coordinator in the health ministry, Dr Herbert Kadama, said preparations for the launch are ongoing.
KAMPALA - Uganda is set to launch lenacapavir, the long-acting injectable HIV prevention drug, on March 25, 2026.
The national pre-exposure prophylaxis (PrEP) coordinator in the health ministry, Dr Herbert Kadama, said preparations for the launch are ongoing.
Dr Kadama further noted that the launch simply means giving the first injection of the drug to a selected beneficiary.
After, the drug will be rolled out in about 300 public and private not-for-profit (faith-based) health facilities across the country.
The development comes after Uganda received the first consignment of 19,200 doses of lenacapavir on February 24, 2026.
According to Dr Kadama, Uganda will receive the drug in a phased manner until all the expected 94,000 doses are delivered.
Breaking down the delivery schedule, Dr Kadama said the second consignment of 7,000 doses is expected in May. In September and December, more batches of 30,000 doses will be delivered, respectively.
Earlier this year, the National Drug Authority approved lenacapavir, manufactured by Gilead Sciences, for use as a twice-yearly PrEP option.
The injectable HIV prevention drug lenacapavir is administered every six months to prevent HIV acquisition among people in high-risk populations.
The categories of people to benefit include adolescent girls and young women aged 15-24, pregnant and breastfeeding mothers, female sex workers, and their male clients. The other targeted populations include fishermen, long-distance truck drivers, and people in multiple sexual partnerships.
Named the “Breakthrough of the Year” by Science magazine in 2024, lenacapavir demonstrated 100% efficacy in reducing HIV infections in early study results.
Lenacapavir, the superior medical innovation, was developed by the United States company Gilead Sciences with support from the US government.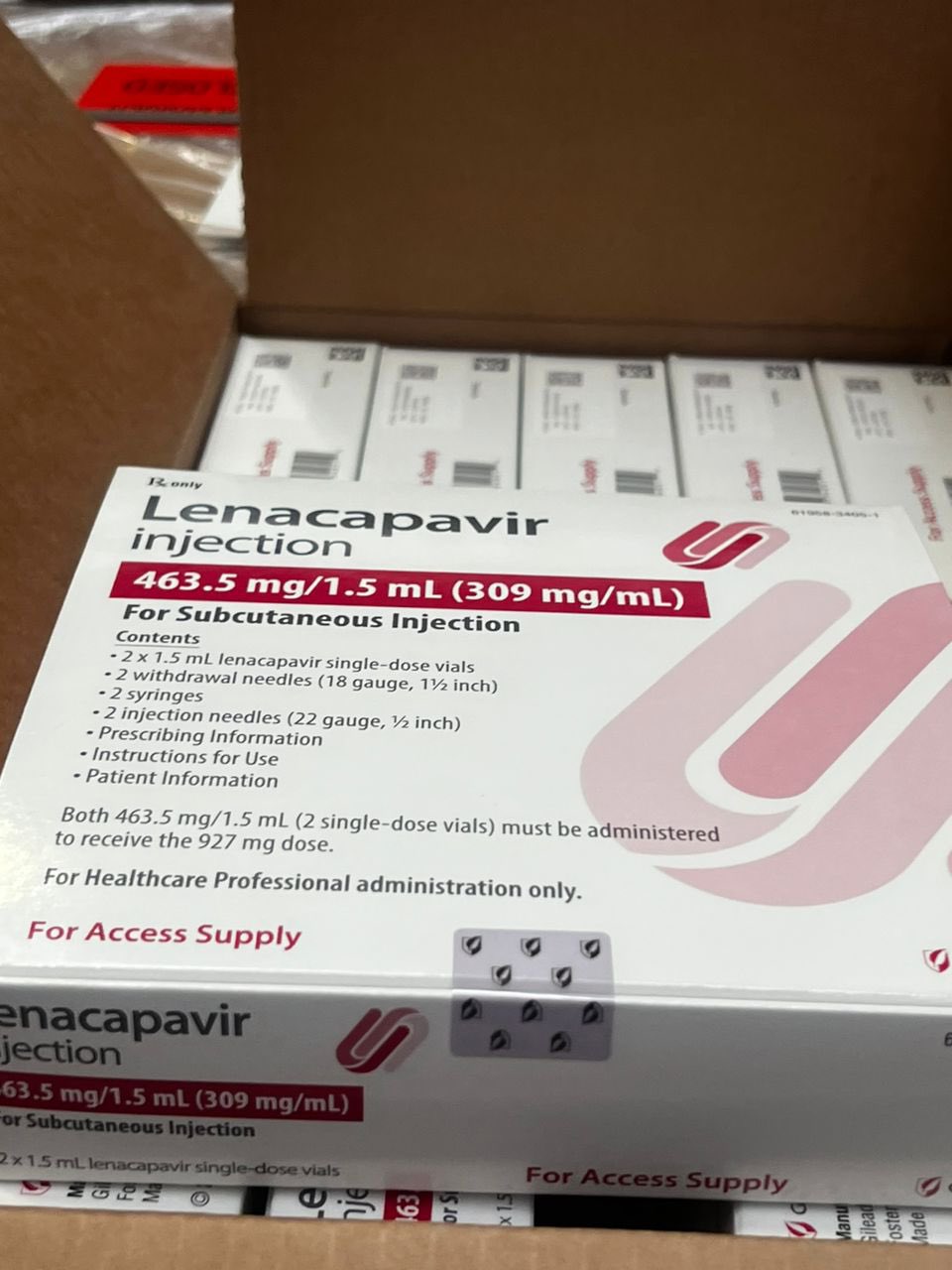
Lenacapavir Injectable HIV prevention drug.
HIV burden
Uganda continues to grapple with high rates of new HIV infections. In 2024 alone, 37,000 new infections were recorded nationwide.
Young women aged 15-24 remain disproportionately affected, accounting for 78% of infections among adolescents. Four out of every five young people newly infected are girls and young women.
However, the overall HIV prevalence has slightly declined from 5.1% in 2023 to 4.9% last year. Annual AIDS-related deaths have also dropped significantly, from 54,000 in 2010 to 20,000 in 2024.
Approximately 1.5 million Ugandans are living with HIV, with about 1.3 million currently on antiretroviral therapy.